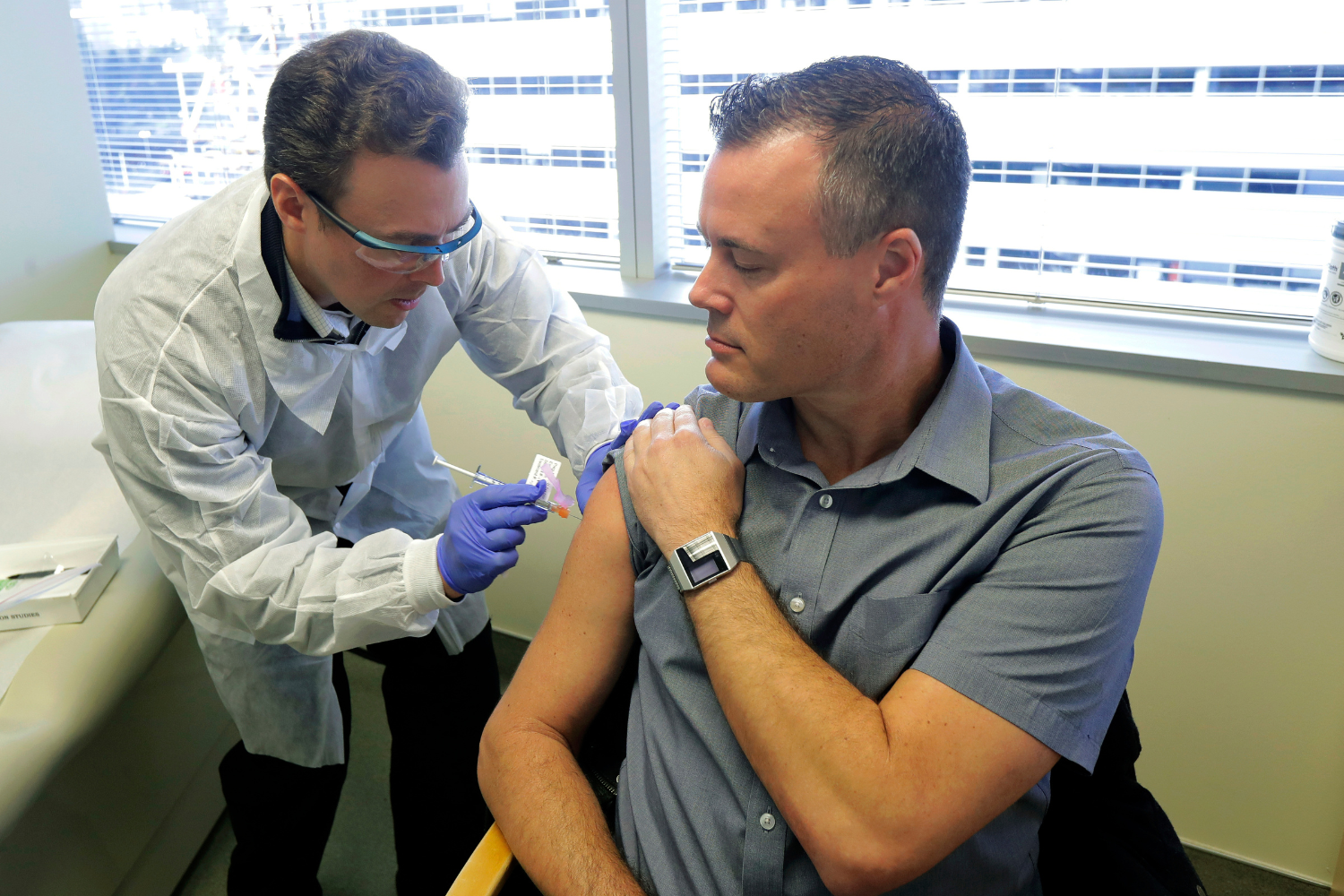
First Man in the US To Get COVID Jab Reveals Side Effects 6 Years Later
Summary
Neal Browning, one of the first people to receive the Moderna COVID-19 vaccine in a trial, shared his experience and the side effects reported after six years. The trial, which began in March 2020, marked an early step in the path to COVID-19 vaccinations but became associated with public mistrust and misinformation over time.Key Facts
- Neal Browning was one of the first participants in the Moderna COVID-19 vaccine trial.
- The trial began on March 16, 2020, in Seattle at Kaiser Permanente Washington Health Research Institute.
- The trial included 45 volunteers, each receiving two doses of the vaccine.
- The trial focused on safety and whether the vaccine could trigger an immune response.
- False claims about vaccine dangers circulated online, leading to public mistrust.
- In 2025, the FDA required warnings for certain heart issues linked to the vaccine for young males.
- An internal FDA memo in 2025 reported at least 10 child deaths "related" to COVID-19 vaccination but lacked detailed evidence.
- The CDC changed its recommendation in 2025 from a blanket vaccination policy to individual decision-making.
Read the Full Article
This is a fact-based summary from The Actual News. Click below to read the complete story directly from the original source.